Top Links
Journal of Pharmaceutics & Drug Development
ISSN: 2348-9782
Review on Transdermal Drug Delivery Systems
Copyright: © 2014 Pang Z. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Related article at Pubmed, Google Scholar
Recently, transdermal drug delivery system (TDDS) has become a more and more important approach to administering drugs. Based on its advantages, which are not achievable by other modes of administration, many researchers are dedicated to the study of it, and have made great progress. Although the skin offers a painless interface for systemic drug delivery, it also presents limitations which are mainly caused by the stratum corneum. In this work, we state the increasingly impact of TDDS, discuss the limitations of it, and last but not least, we highlight the methods for overcoming these limitations by using permeation enhancers, microneedles, iontophoresis.
Keywords: Transdermal drug delivery system; Permeation enhancers; Iontophoresis; Microneedles
During the past decades, intensive studies have focused on the technologies in drug delivery, and with the rapid developments and explorations in technologies, traditional drug delivery means are being replaced by the more effective and advanced ones. The creation of transdermal drug delivery system (TDDS) has been one of the most sophisticated and innovative approaches of drug deliveries. The transdermal drug delivery system has attracted considerale attention because of its many potential advantages, including better patient compliance, avoidance of gastrointestinal disturbances, hepatic first-pass metabolism and sustained delivery of drugs to provide steady plasma profiles, particularly for drugs with short half-lives, reduction in systemic side effects and enhanced therapeutic efficacy [1-6].
Despite these advantages, most of the transdermal candidates have low permeability. The drugs administered across skin should have the three constraining characteristics: appropriate partition coefficient, low molecular mass (<500Da), and small required dose (upto milligrams) [3]. The limitations of transdermal drug delivery are caused by skin which protects against and is impermeable to foreign molecules. The human skin is consisted of two main layers: the layer of epidermis and the layer of dermis. Stratum corneum is the epidermiss outermost layer that composed of stratified keratinocytes, multiple lipid bilayers of ceramidas, fatty acids, cholesterol and cholesterol esters. Stratum corneum provides an extremely effective physical barrier for the control of drug penetration [2,7-10]. Therefore, attempts to overcome this skin barrier is presently an important area of pharmaceutical and toxicological research. The techniques that weaken the barrier have included permeation enhancers [11-43], microneedles [44-68] and iontophoresis [69-84].
In the last 50 years, a large number of chemical permeation enhancers (CPEs) which are defined as substances that interact with the major constituents of skin barrier, stratum corneum, to promote penetration of drugs into skin.
The ideal enhancer should have the following conditions:
1. Non-pharmacological activities.
2. Nontoxic, non-allergenic, and non-irritating.
3. Rapid-acting with predictable and reproducible activity.
4. When removed from the skin surface, the penetrability of the skin should recover immediately.
5. Cosmetically acceptable with suitable skin feel [11].
The part of penetration enhancer in topical formulations has been becoming significantly and undoubtedly, and they would permit the delivery of broader classes of drugs through the stratum corneum in the future.
From the above mentioned, we can know that the skin provides such a formidable obstacle to the delivery of most drugs, a variety of different chemical permeation enhancers have been tested to enhance transdermal penetration. Immense amounts of research during the past two decades has led to the formulation of some different classes of penetration enhancers, including surfactants (for example, sorbitan trioleate), fatty acids/esters (for example, lauric acid), terpenes (for example, menthone), azone-like compounds (for example, azone), and solvents (for example, ethanol and dimethyl sulphoxide) (Table 1) [1,6,12-15].
Fatty acids for the use of skin permeation enhancers: Fatty acids contain a aliphatic hydrocarbon chain and a terminal carboxylic acid group. Because the number, position, and configuration of double bonds of their aliphatic chain, which is either saturated or un-saturated, are visible different, and the aliphatic chain may have branching and other substituents, fatty acids have different characteristics and enhancing effects [16,17].
A study of Florence et al. indicates that with the increasing of the carbon chain length, the lipophilicity of fatty acids also increases. So long-chain fatty acids have a much higher affinity to lipids in the stratum corneum, while short-chain fatty acids have insufficient lipophilicity which goes against the skin penetration [18].The work of Guy et al. shows that the stratum corneum of the human skin is a lipophilic organizational structure and that compounds of high lipophilicity tend to remain in the SC, preventing drugs to transport to the deeper tissues [19]. Evidences from many studies have confirmed that the enhancing effects of fatty acids were most significant for C10 to C12 fatty acids [20,21].
In addition, the enhancing effects of fatty acid was also influenced by the bond saturation. Some studies have demonstrated that the unsaturated long-chain fatty acids have been shown a greater enhancer activity when compared to saturated fatty acids of the same chain length. This is mainly attributed to the higher disrupting nature of the unsaturated long-chain of these fatty acids that would result in a higher magnitude of lipid disruption [22,23].
Last but not least, the enhancement effects of fatty acids on penetration through the stratum corneum are also dependent on the concentrations of fatty acids. Some studies have shown that the skin permeation of Meloxicam through human skin increased when the concentration of oleic acid increased from 0.4 to 1 %.
Terpenes for the use of skin permeation enhancers: Terpenes are a series of volatile oil from natural sources or laboratory designed, and consist of repeated isoprene (C5H8) units, in a head-to-tail orientation to form linear chains or rings, which is the basic chemical structure of terpenes [24,25].
Terpenes, as a kind of clinically acceptable penetration enhancers have the following advantages:
1) When compared to other synthetic skin penetration enhancers, terpenes are generally less toxicity with low irritancy. And quite a few terpenes have been classified as generally regarded as safe by the Food and Drug Administration (FDA) [26-30].
2) Reversible effect on the lipids of stratum corneum [31].
During the past two decades, the satisfactory performance of terpenes in enhancing the permeation of drug through the skin has been intensively revealed. Terpenes can increase skin permeation by interacting with SC lipids and keratin, and increasing the solubility of drug into SC lipids [26]. There are some main factors that play an important role in the permeation enhancing activity of terpenes:
1. The chemical structures of terpenes: It has been commonly observed that terpene stereoisomers can affect their enhancing activity. Some researcher have reported that the (-) enantiomer of a terpene is more effective than the corresponding (±) racemate or the (+) isomer. Futhermore, some terpenes with a minimal degree of saturation are more effective enhancers to deliver the hydrophilic drugs [32,33].
2. The lipophilicity of terpenes: High lipophilicity is an important structural feature for terpenes; accordingly, hydrocarbon, nonpolar terpenes, such as limonene, are more effective to promote the transdermic absorption of lipophilic drugs than oxygen-containing polar terpenes and vice versa [34,35].
3. The physical form of terpenes: From some studies, we can know that the liquid terpenes have a greater ability to form hydrogen bonds with intercellular lipids of SC, so liquid terpenes can produce better enhancing effects than the solid ones [36].
Azone-like compounds for the use of skin permeation enhancers: Azone, see Figure 1, is a highly lipophilic substance with a logP around 6.2, and it can be compatible with many types of organic solvents, such as alcohols and propylene glycol. Besides, Azone also has low irritancy and very low toxicity [37].
In the last few years, Azone is one of the most investigated enhancers; it is more effective for hydrophilic drugs than lipophilic drugs. And as a skin permeation enhancer, the efficacy of Azone appears to be dependent on its concentration, and surprisingly, it is more effective at lower concentrations, being employed typically between 0.1 to 5%. However, the lag time of Azone was very high, which is from 2h to 10h, but its effects can last for many days [38].
Azone increases the ability of drugs to permeate the skin by interacting with the lipid domains of the SC, disordering lipid bilayers, disrupting their packing arrangement, and increasing lipid solubilization [38].
Chemical penetration enhancers can dramatically increase skin permeability by reducing the resistance of the skin to drug diffusion [39-41]. Skin permeation enhancers may exert their effects by one or a combination of the following several possible mechanisms:
1. Interaction with the intercellular lipid matrix (especially cholesterol and ceramides), disrupting the highly ordered lamellar structure.
2. They can interact with intracellular protein (influencing the conformation of keratin in the corneocytes or proteins in desmosomes) to promote permeation of drugs through the corneocyte layer.
3. CPEs can improve the partition of the drugs, increasing the concentration of the drug in the skin.
4. CPEs can dissolve the skin-tissue components [42,43].
Recently, microneedles (MNs) have gained more and more attention as the excellent candidates for transdermal delivery. MN arrays are composed of multiple micron-sized projections which are typically assembled on one side of a supporting base or patch. These microprojections generally range from lengths as short as 25μm to those as long as 2000μm, so that they can create little holes in the stratum corneum without pian and become the effective way to enhance the delivery of therapeutic molecules and macromolecules complexes across biological membranes including mucosal tissue, and skin, sclera [44-46].
Microneedles can be fabricated from different kinds of materials, such as silicon, silicon dioxide, metals such as stainless steel, palladium, nickel and titanium, glass, ceramics and polymers, and they differ in shape and in length. MN have been produced in various geometries. These microstructure geometries can be in the form of needle-like (most common MN geometries which can be sharp-, tapered-, conical- or bevel-tipped), microblades, and blunt-projections or shaped in an arrow-head [47-51].
In addition, MNs as a parenteral route of administration could provide the relatively easy and patient-friendly administration of therapeutics with potential efficacy at low cost. And some studies have shown that MNs can penetrate the skin, cross the stratum corneum into the viable epidermis, and reside primarily in the dermal layer, avoiding contact with blood vessels and nerve fibres. So MNs can provide a pain-free, minimally invasive ways to delivery both small and large molecular weight without bleeding at the application site (Figure 2) [54].
Over the last decade, extensive experiments about MNs has been carried out with the use of a wide range of techniques. It is worth mentioning that the effect of MNs has been demonstrated in in vitro, ex vivo and in vivo experiments.
MN arrays can be classified two different types: solid microneedles (Figure 3a,b and c) and hollow microneedles (Figure 3e and f) [51-53], which are commonly described in the literature. As the names imply, the two types of MNs have been used by different approaches for the delivery of therapeutics across the skin.
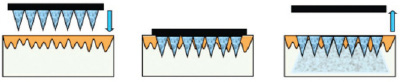
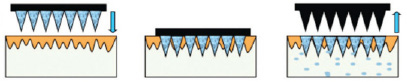
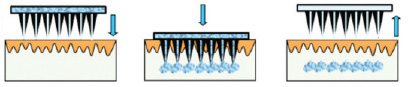
Different types of MNs have different ways of drug delivery. There are four general means to delivery drug by microneedles, as schematically depicted in Figure 4.
Drug delivery by solid microneedles: There are three main approaches which can be used to deliver drugs into the skin by solid microneedles, including the "poke and release" approach, the "poke and patch" approach, and the "coat and poke" approach, as schematically shown in Figure 4a, b and c.
In an early phase of research on microneedles, an array of solid microneedles made up of silicon wafers was used to pierce through skin to overcome the barrier caused by the stratum corneum. Subsequently, the microneedles were removed and transient microchannels were created in the skin upon which a conventional patch containing the drug formulation was applied onto the treated skin surface so that the drug can diffuse to the formed microchannels. This approach is named as "poke and patch", which is the first strategy of MN-mediated transdermal drug delivery [55-57].
Some studies have reported that the permeability for different drugs was increased by "poke and patch" approach up to four orders in magnitude. And for the "poke and patch" approach, the key is that the micropores should stay open conditions during the drug application period. However, it has been demonstrated that the micropores generated by microneedles can keep open conditions until at least 72h and this approach may not be applicable, because longer opening times can be along with the risk of infection [58-61].
Subsequent researches in microneedles result in the development of a "poke and release" approach. The second approach for (Trans) dermal drug delivery features a modulated release of the drugs from the microneedles by using a variety of available materials, such as polymers and polysaccharides, which serve as a drug depot and can be either slowly degraded or dissolved after administration. It means that, the microneedles must remain on the application site until the drug is totally released. The administration of a large amount of drug is still not feasible by the "poke and release" approach, which lead to the development of "coat and poke" approach [62].
The further current approach for (Trans) dermal drug delivery by solid microneedles is via the "coat and poke" approach. Solid microneedles are coated with drug solution by dip-coating technology, containing DNA, RNA, proteins, hydrophilic and hydrophobic low-molecular-weight drugs, inactivated pathogens, and particles, etc. After the skin was pierced, the drug can be completely released. However, a limited quantity of drugs could be coated over the microneedles (only about 1 mg) because thick coatings can decrease sharpness of microneedles, resulting in a very low skin delivery efficiency. Besides, uniform coating in the process is the key, which can be achieved by optimizing the experimental conditions. Lastly, during the process of coating, the challenge is the relatively high loss of drug. In order to solve the problem, multiple methods of coating and coating devices have been invented during the past decade [63-65].
Drug delivery by hollow microneedles: Hollow microneedles deliver drugs via a more effective approach (Figure 4d). This approach was known as"poke and flow". After the microneedle system penetrates into the skin, the drug solution was allowed to flow through the pore from the reservoir and release into the skin. The most important advantage of hollow microneedles over the approaches above-mentioned is the possibility to facilitate fluid flow by the driving forces, such as pressure, thus a large amount of drug can be administered by hollow microneedles, thereby allowing higher rates of drug delivery [66,67]. In general, to prevent blockage of channel and insert the microneedle easily, the pore is kept alongside walls instead of at the centre.
In order to improve the permeability of insulin unilamellar, Chen et al. researched the delivery efficiency of the drug through hollow microneedles combined with iontophoresis so the delivery of drug can be in a controlled way by regulating the current. The current may be turned on and off by the patient based on their own needs and desires. Even though the combination of hollow microneedles and iontophoresis can increase the delivery efficiency, this leads to skin irritation when a constant current is used [68].
Although the design of the microneedle is more effective to achieve drug delivery compared with other approaches, and the microneedle can be used to deliver drugs either at a local site or systemically and do not cause any pain or irritation, the disadvantages of microneedle system are that: (1) a small amount of drug can be given (less than 1mg); (2) the accuracy of dosage may be less than hypodermic needles; (3) the hollow microneedles must be not blocked and broken off; (4) repetitive injection may destroy the veins; (5) the external environment, such as hydration of the skin, could affect delivery.
Iontophoresis, which is defined as "the application of a small electric current (0.5 mA/cm2 or less) with a low voltage, to drive ionic and polar molecules across the skin and into the tissues", has been used to improve the transdermal delivery of drugs, such as neutral and charged molecules, low and high molecular weight drugs namely phenobarbital, ranitidine and zidovudine. Basically, Iontophoresis consists of two electrodes: a cathode and an anode. Commonly, the small electric current is driven from an external supply into the skin at the anode, and back out at the cathode (Figure 5). Direct current iontophoresis with a constant current approach is the most common form of iontophoretic transport. Alternating current has also been employed in transdermal iontophoretic drug delivery. Some reports has suggested that alternating current compared to direct current can reduce skin irritation, eliminate potential electrochemical burns during long iontophoresis application. Under the circumstances when iontophoresis is used, the direct current or alternating current electric field reduces skin electrical resistance by the mechanism of electroporation. This electroporation effect has been found to dramatically increase as the voltage is increased. Because iontophoresis has many advantages, such as ease of application, high efficiency, safety and miniaturized instrument, it is known as a promising approach to improve transdermal drug delivery [68,69].
However, several limitations and disadvantages have been associated with the use of such approach, including stability, loadability, handleability. Besides, chemical and thermal stability of the reservoir and the electrode is one of the considering problems.
The mechanisms of transdermal iontophoresis include electroosmosis (electric field induced solvent flow), electromigration (electric field charge interaction) and electroporation (increasing the porosity of skin due to electric field).
Electroosmosis: Transdermal iontophoresis can enhance the transport of ionic drugs by the imposed electric field or voltage. The electric field imposes an additional force, and then drives the ion through the skin more efficiently than the pure diffusion or "passive" transdermal drug delivery. In general, if the drug is not ionogenic, there is no direct interaction between the drug and the electric field, so drug delivery should not be improved. However, some studies have demonstrated that electroosmosis can cause bulk solvent flow, which is suggested as the possible mechanism for the promoted permeability of neutral species.
Electroosmosis refers to the relative movement between liquid (usually water) and the fixed solid phase in the electric field. Electroosmotic flow is referred to bulk flow of the solution or volume flow, which occurs in a wide variety of membrane systems containing fixed charges. This flow is not diffusion, which has nothing to do with concentration gradients. The direction of flow is always in the direction of counterion flow, which may be against or with the current flow, depending on the nature of the membrane systems [70-72].
Electroporation: Electroporation utilizes short, high-voltage electrical pulses (50 to 2000V) to create temporary pores or aqueous pathways within the SC and promote transdermal drug delivery, which are efficient for DNA or large hydrophilic drug molecules delivery. The reason is that electroporation results in fluidization of lipids and phase transition because of joule heating and higher localized temperature (over 60 °C). The main modes of electroporation are diffusion and electrophoresis. Under the action of high-voltage electrical pulses, the transdermal transport of drugs occurs via localized regions of the stratum corneum. In these regions, the small molecule drugs can be transported via the transcellular route in the condition of short high-voltage electrical pulses, but the intercellular route possibly with appendageal pathway is adopted when lower voltage and prolonged pulse duration are used.
Electroporation can be used together with chemical enhancers such as magnesium chloride, calcium chloride, calcium bromide, heparin, urea, anionic phospholipids and sodium thiosulfate. These chemical enhancers such as magnesium chloride can interact with the lipid bilayers of SC and overcome the skin barrier with slow recovery under the influence of electroporation. However not all the chemical enhancers can disrupt the lipid lamellae. Instead, they insert themselves into the aqueous pathways or pores, and then stabilize the transient disruption created by electroporation. The combination of chemical enhancer and electroporation will enlarge the aqueous pores and prolong the lifespan of them [73-76].
Electromigration: The electroosmosis can be useful for both charged and neutral molecules whereas electromigration is primarily used to drive the charged molecules across the skin. Under the influence of electric current, the appendageal pathway predominates when the ions tend to travel the path which offers the least resistance. On the other hand, the non-appendageal pathway has also been identified as an important player in iontophoretic delivery when the intercellular route is concerned. The intercellular route consists of polar regimes in lipid lamellae. When electrical current is imposed, these polar aqueous pores are formed as a result of flip-flop movement of polypeptide helices in the stratum corneum. They confer an alternative pathway for drug transport [77-79].
Nowdays, it has been confirmed that MN will result in creating of aqueous pathways of low electrical resistance. The synergistic effect of iontophoresis and MN technologies can lead to an increased rate of transdermal delivery for a range of drug molecules with the benefit of precise electronic control. In order to date the combination of iontophoresis and MNs, some researchers have focused on either the use of an electric stimulus to promote drugs to move through the central bore of hollow MNs or the use of non drug-loaded solid MNs (used to merely puncture the skin) before the application of an electrically conducting drug formulation. In these cases, the central bore of the hollow MNs may become blocked and the pores created by the solid MNs could be likely to close, and the use of iontophoresis may stimulate drugs to move. The use of hydrogel- forming MN or drug-loaded dissolving polymer MN systems may overcome some of the above mentioned problems and thus allow the combination of MN and iontophoresis to enhance transdermal delivery of a wide range of drug molecules [80-84].
Transdermal drug delivery is an alternative and promising way to systematic administration of drugs on account of the validated therapeutic effect, safety, low cost and convenient use. To over the barrier and improve transdermal efficiency, these methods mentioned above have been deeply investigated and tested in a number of humans clinical trials and they enable patient-controlled dosing and more complex delivery profiles than passive transdermal drug delivery systems.
This work was supported by the Foundation of Jinan Science and Technology Development Program (201303055). This project was supported by Project of Shandong Province Higher Educational Science and Technology Program (J14LK61).
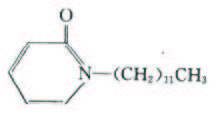 |
| Figure 1: The structure of laurocapram (1-dodecylazacykloheptan-2-one, laurocapram,6) |
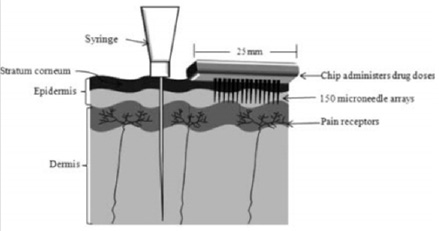 |
| Figure 2: Reference of the comparison of MN arrays and ordinary syringes |
 |
| Figure 3: The images of solid microneedles (a,b and c) and hollow microneedles (e and f) |
 |
| Figure 4a: "poke and patch" approach |
 |
| Figure 4b: "poke and release" approach |
 |
| Figure 4c: the "coat and poke" approach |
 |
| Figure 4d: "poke and flow" |
| Figure 4: Reference schematic diagram of the approaches for drug delivery by different designs of microneedles: (a) 'poke and patch' using solid microneedles; (b) 'coat and poke' using coated solid microneedles; (c) 'poke and release' using polymeric microneedles; (d) 'poke and flow' using hollow microneedles |
 |
| Figure 4a: "poke and patch" approach |
 |
| Figure 4b: "poke and release" approach |
 |
| Figure 4c: the "coat and poke" approach |
 |
| Figure 4d: "poke and flow" |
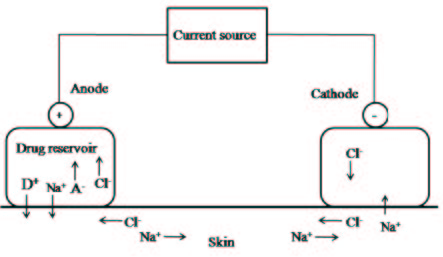 |
| Figure 5: Reference schematic diagram of the sketch of iontophresis using an Ag/AgCl electrode system: Na+ = sodium ion in drug reservoir, Cl− =chloride ions in drug reservoir or from AgCl electrode, A− = anionic species of drug, D+ = cationic species of drug. |
| Types | Examples |
|---|---|
| Surfactants | Sorbitan monopalmitate, Sorbitan trioleate, Cetyl trimethyl ammonium bromide |
| Fatty acids/esters | Alkanoic acids, oleic acid, lauric acid, capric acid, Cetyl lactate, butyl acetate, isopropyl myristate |
| Terpenes | Nerolidol, Farnesol, Carvone, Menthone |
| Azone-like compounds | Azone (Laurocapram, 1-dodecylazacycloheptan-2-one), 1-alkyl- or 1-alkenylaza cycloalkanones |
| Solvents | DMSO, ethanol, 1-octanol, 1-hexanol |
| Table 1: Types of chemical penetration enhancers | |






































