Top Links
Journal of Case Reports and Studies
ISSN: 2348-9820
Hypoglossal Schwannoma: A Rare Case Report
Copyright: © 2016 Kumar SN. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Related article at Pubmed, Google Scholar
Hypoglossal schwannomas are rare cranial base neoplasm arising from schwann cells of the XIIth cranial nerve. We report a case of 45 year old lady with history of headache for 2 years and difficulty in swallowing for 1 year. On evaluation, she was found to have left hypoglossal paresis, atrophy of tongue muscles on left side and a smooth oropharyngeal bulge on left side. Imaging showed a well-defined hypoattenuating mass in the left parapharyngeal region with widening of hypoglossal canal and displacement of carotid artery anteriorly. The tumour was excised into and was found to be arising from the left hypoglossal nerve. The histopathological report confirmed the diagnosis of hypoglossal schwannoma.
Keywords: Schwannoma; Hypoglossal nerve; Parapharyngeal tumor
Schwannomas are benign, slow-growing neoplasms arising from the myelin-producing Schwann cells in the peripheral sensorimotor nervous system. More than 90% of cases are vestibular schwannomas [1]. Hypoglossal nerve is a pure motor nerve. The schwannomas arising from hypoglossal nerve is very rare and accounts for only around 100 cases of hypoglossal schwannoma in english literature [2,3]. Hypoglossal schwannoma are usually found to originate intracranially but can also extend extracranially through the hypoglossal canal in a ‘dumb bell’ shape or can arise purely from the extracranial portion of the XII nerve.
A 45 year old lady, presented with history of headache for 2 years and gradually progressive swallowing difficulty for 1 year. On evaluation, she was found to have a smooth bulge 3x4 cm involving left lateral wall of the oropharynx, pushing the tonsillar fossa medially. There was a deviation of tongue towards left side and hemiatrophy of the tongue on left side. Fibrillations were also noted in the atrophied tongue. A 2x3 cm smooth mass, which was non-tender and firm in consistency, could be palpated below and behind the left submandibular gland. No other cranial nerve palsies were noted .There were no other swellings palpable in the neck.
A contrast enhanced CT scan was taken and it showed a well-defined hypoattenuating mass of 52x47x24 mm in the left parapharyngeal region (Figure 1) extending from the skull base with widening of hypoglossal canal (Figure 2) and displacement of internal carotid artery anteriorly and medially. External carotid artery was displaced laterally. Medially it was abutting the mucosa of left oropharyngeal wall. An image guided FNAC was done was s/o spindle cell neoplasm.
The mass was excised trans-cervically, after removing the left submandibular gland for adequate exposure. Left hypoglossal nerve was hypertrophied in the distal part and was traced upwards towards the tumour. A smooth elliptical mass of 5x 3x 2 cm was present in continuity with the hypoglossal nerve in its proximal part and was coming out of the hypoglossal canal widening the same. The tumour was dissected out from the carotid sheath and surrounding structures. There were no complications like haemorrhage and there was no need for multidisciplinary approach.
Histopathological picture was suggestive of spindle cell neoplasm from peripheral nerve sheath cell- schwanomma (Figure 3). Immunohistochemical examination showed positive for S-100 protein. Post operatively she had features of left hypoglossal palsy.
Schwannomas originate from the Schwann cells surrounding the peripheral nerves, including cranial nerves III to XII. More than 40% of non-syndromic benign solitary schwannomas are located in the head and neck region [4]. The most frequently involved cranial nerve is the eighth cranial nerve and vestibular schwannomas account for 8% of intracranial tumors [5]. Non-vestibular schwannomas are rare and constitute <0.5% of all intracranial tumors [6]. Schwannomas derived from lower cranial nerves are usually seen in the cervical region, jugular foramen, and hypoglossal canal. Most of the skull base schwannomas arise from the sensory nerves; purely motor nerve–associated schwannomas are very rare and often associated with von Recklinghausen disease [7]. The parapharyngeal space is the most common site of tumor origin (31%), followed by the neck (23%), skull base (19%), sinonasal cavity (15%), middle ear (8%) and posterior pharynx (4%) [8].
Most of the patients presented with symptoms secondary to mass effect or nerve deficit and the commonest presentations were neck mass (27%), dyspnea/dysphonia (15%), nasal obstruction/epistaxis (12%), odynophagia/dysphagia (12%) and shoulder/upper extremity pain (8%) [8].
Hypoglossal schwannomas were reported for the first time by De Martel in 1933 [1]. They represent only 5% of all nonvestibular schwannomas, with approximately 110 cases reported in the English literature to date.Of these cases, the vast majority of cases originate intracranially, with or without extracranial extension [9,10]. According to Kaye et al. the hypoglossal schwannomas can be divided into three types:
Type A: Intracranial (31.5%)
Type B: Dumb bell shaped (50%) [11]
Type C: Extracranial (18.5%) [12,13]
According to this classification, our case is type c which is comparatively rare.
Nonaka et al. also classified the lesions involving the hypoglossal canal as: Type A: Intradural tumor; Type B: Transdural and extradural dumbbell-shaped tumors; and Type C: Extracranial base tumor [14,15].
Schwannomas arising exclusively from the extracranial segment of the hypoglossal nerve are exceedingly rare. The first case was reported by Slaughter et al in 1949 [9]. In a review of the literature by Cavalcanti et al. in 2011, only 36 peripheral extracranial hypoglossal schwannomas were reported [16].
Hypoglossal schwannomas seems to have a female predilection (2.5:1) and mean age at diagnosis was 46 years [10,16]. These figures are in accordance with our case. The clinical presentation can vary according to the site and nerve of origin. Most often, earliest feature as reported is wasting of the tongue muscles (25-94%).Hypoglossal nerve weakness is a result of expansion and streching of the nerve fibres during the tumour growth [9,10,16]. Kubota et al. suggested that motor cranial nerves are more resistant to compression as they have a thick myelin sheath [17].
Intra cranial tumours may present with suboccipital head ache exacerbated by neck movements. Large tumours may present with cerebellar or brainstem signs. Extracranial tumours usually present with neck swelling [8,18].
Imaging is required for the pre-operative evaluation. MRI is useful in soft tissue characterization, while CT scan is useful to assess the relation with skull base. An image guided FNAC can be attempted as metastatic lymphnodes and tumours from deep lobe of parotid gland constitute major differential diagnosis [19,20]. Complete surgical excision is the treatment of choice and the chance of recurrence is very less. According to the site and extent of the lesion, surgical approach has to be planned and a multidisciplinary team may be required for the complete excision.
These tumors are benign slow-growing tumors; thus complete resection offers cure. The morbidity and mortality is related with the brainstem compression and associated cranial nerve palsies [21-23]. Steriotactic Radiosurgery is an alternative treatment that has emerged recently and can be used as an adjuvant or in the salvage setting or for very small tumours especially when they are adjacent to critical structures. Though there is a theoretical risk of 0.005 to 0.1% for secondary malignancy or malignant transformation, but so far not many cases are reported [24].
Neurogenic tumours are frequently seen in the parapharyngeal space; but a schwannoma arising from hypoglossal nerve is very rare. A careful clinical examination and preoperative imaging will lead to the diagnosis and thus proper surgical planning; which is essential to prevent complications and recurrences.
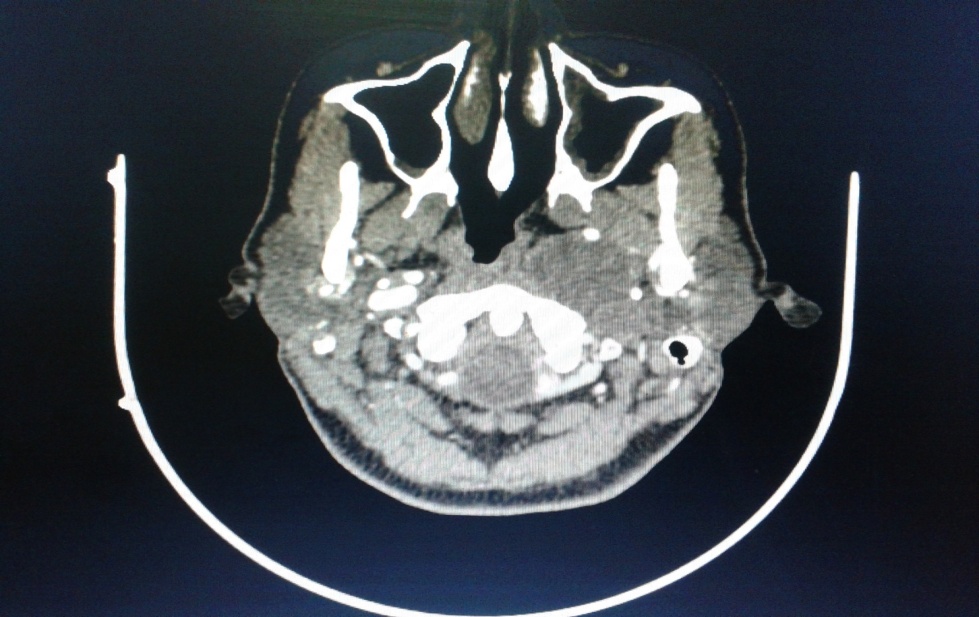 |
| Figure 1: Well-defined Hypoattenuating mass of 52x47x24 mm in the left parapharyngeal region |
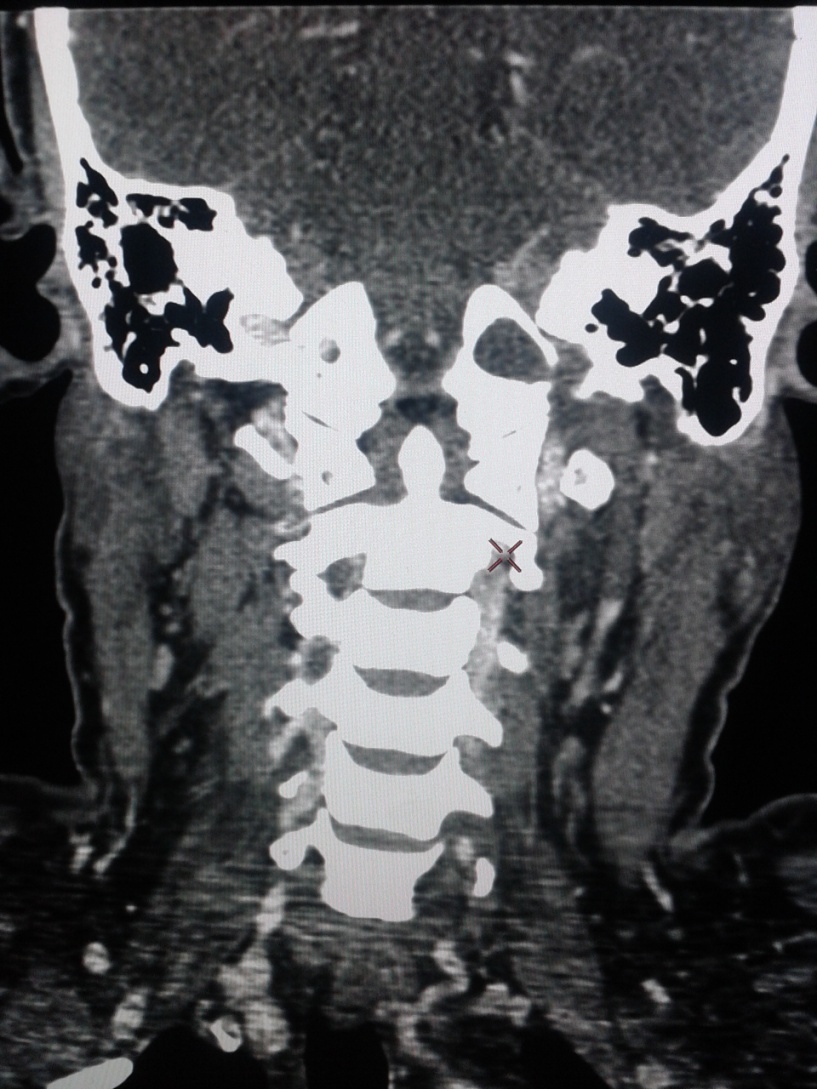 |
| Figure 2: Widening of left hypoglossal canal |
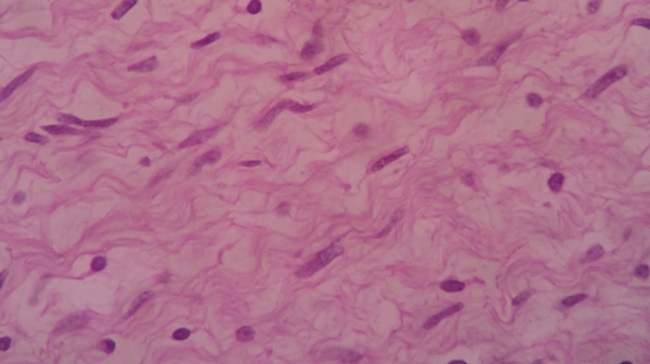 |
| Figure 3: Eosin and hematoxylin stained spindle cell proliferation |






































