Top Links
Journal of Surgery and Operative Care
ISSN: 2455-7617
Predictors of Stress Fracture Susceptibility in Arab Female Military Recruits during Combat Training: A Preliminary Study
Copyright: © 2016 AlJawder A. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Related article at Pubmed, Google Scholar
Introduction: Military recruits and athletes regularly engaging in vigorous physical activities are at increased risk for sustaining lower limb overuse injuries. Stress fractures represent up to 20% of all overuse injuries sustained by such populations. Risk factors of stress fractures have been recently shown to vary between different ethnic and demographic groups. The aim of this study is to evaluate risk factors of stress fractures among Arab female military recruits engaged in basic military training.
Methods: A prospective study was conducted on 108 Arab female military recruits entering 6-months Basic Military Training program in 2014-2015. During the program’s orientation and pre-entry medical examination, information on age, weight, height, hemoglobin, alkaline phosphatase, serum calcium and vitamin D levels were collected. Participants were also administered a questionnaire pertaining data on marital status, age of menarche, oral contraceptive use, menstrual period pattern and use of vitamins and other supplementations. Participants sustaining lower limb injuries were further evaluated with plain radiographs and bone scans.
Result: 100 females were included in the study, of which 17 (17%) were referred to the orthopedic clinic for further evaluation. A total of 8 participants were diagnosed with stress fractures (8%). Stress fractures of the tibia were most common (87.5%), followed by femur (12.5%). Age of menarche and weight were identified as statistically significant risk factors for sustaining stress fractures. Although vitamin D deficiency was seen in 78% of participants, it did not significantly increase the risk for stress fractures.
Conclusion: Identifying risk factors for stress fractures is important in attempting to reduce their incidence. Risk factors for stress fractures vary between different ethnic populations. This is the first preliminary study to investigate risk factors for stress fractures in Arabs, and could serve as a basis for future stress fracture research in the region.
Keywords: Bone health; Female athletes; Risk factors; Stress fracture; Vitamin D
Bone, due to its mechanical properties, has the ability to withstand certain strain magnitudes during mechanical loading, beyond-which it fails, this is known as the point of ultimate strength. Fatigue failure occurs when repetitive loading leads to bone failure with a magnitude below its ultimate strength. In other words, the amount of strain needed for a bone to fail is reversely proportional to the rate of loading cycles. In conditions where either bone loading or the rate at which it is loaded (or both) are increased, a bone’s endurance threshold is eventually breached this damage is termed a stress fracture.
Military recruits and athletes regularly engage in vigorous training. Repetitive loading to their lower extremities places them at an increased risk for sustaining lower limb overuse injuries. Of all overuse injuries sustained by athletic populations, stress fractures represent up to 20% [1]. A large-scale study conducted on US Army Basic Combat Training recruits reported a stress fracture incidence of 19.3 and 79.9 stress fractures/1000 recruits in males and females, respectively [2]. Higher incidences, reaching up to 28.9%, were also reported [3].
The aim of this study was to evaluate risk factors for stress fractures and to assess their impact in increasing susceptibility to stress fractures amongst Arab female military recruits. Although risk factors for stress fractures have been previously identified, differences in race and ethnicity have been shown to significantly influence the magnitude of the variable’s effect. Bone research in multiethnic populations demonstrated differing bone quality, response to injury and risk factors for fractures between races [4,5]. Therefore, the importance of identifying ethnicity as a significant variable in studying bone health has been recognized [6].
Risk factors for stress fractures can be categorized as extrinsic and intrinsic factors, in other words modifiable and non-modifiable factors, respectively. Whilst modifiable factors have been examined extensively in literature, recent trends are towards individualizing preventative measures according to non-modifiable factors. More preventative measures could be taken for individuals at higher risks for stress fractures due to non-modifiable factors. Extrinsic risk factors are variables that are not related to the individual participant and they increase the likelihood of injury, whereas intrinsic risk factors are characteristic variables specific to each individual candidate. However, in order to prevent stress fracture occurrence, a risk factor associated with injury must be modifiable [7]. Therefore, classifying the known intrinsic and extrinsic factors into either modifiable or non-modifiable did not only show relevance in injury prevention, but it has also shown clinical benefits [8].
The following prospective study was approved by the research and ethics committee of Bahrain Defence Force (BDF) military hospital. The population sample included all female military recruits aged 18-33 enrolling in a military training program from September 2014 till February 2015. Participants, chosen at random, were briefed on the study and written consent was obtained during the first week of the program.
Military recruits who fail to successfully complete the 6 months of basic training program, for reasons other than attaining a stress fracture, were excluded from this study.
All recruits were compliant with the medical requirements of Bahrain Defence Force military, having previously passed a mandatory medical check-up by training doctors prior to starting the basic training program (BTP). The medical check-up included documentation of age, weight, height, haemoglobin level, alkaline phosphatase level, and serum calcium and vitamin D levels; amongst others. These data were collected into a safe password-protected excel sheet. Additionally, each participant was administered a questionnaire pertaining data on marital status, age of menarche, oral contraceptive use, menstrual period pattern and use of vitamins and other supplementations.
The recruits were there after followed through the six-month period of the training program for occurrence of any musculoskeletal injuries, with particular focus on stress fractures. Military training doctors were the first to assess injuries; they were to refer participants with symptoms of fractures and/or stress fractures to the BDF hospital, where senior orthopaedic residents further evaluated participants. The training officer in-charge was responsible for documenting details of the injuries for each recruit, including date and site of injury, initial and final diagnoses; and patients outcome.
Subjects who complained of persistent bone pain were followed up in the orthopaedic clinic. Diagnosis of a stress fracture was based on: a) clinical presentation pain of sudden onset without previous trauma which is exacerbated and worsened by repetitive physical activity, a confirmatory plain radiograph (X-ray) and followed by a SPECT bone scan (e.cam Signature Series; Siemens Healthcare) (Figure 1 and 2).
Anthropometric measurement: Anthropometric measurements of weight and height were obtained from medical records. BMI was then calculated by dividing the weight in Kg by the squared height in meters. Findings were displayed in a table and plotted in a line graph.
Laboratory investigations: Serum levels of haemoglobin, alkhaline phosphatase, calcium and vitamin D were obtained for each participant prior to commencing the basic training program.
Questionnaire Measurements: Prior to starting the basic training program, participants were administered a paper questionnaire. The questionnaire included basic demographic information and presence of risk factors and co-morbidities. Risk factors included reproductive and menstrual cycle history, such as age of menarche, date of last menstrual cycle, frequency of the cycle/annum, marital status, gravidity and use of oral contraceptive pills (OCPs). Additionally, the questionnaire addressed physical activity and exercise behaviour prior to BTP. The questionnaire also assessed the participants’ diet (dairy food consumption) and use of vitamin D and calcium supplements. Finally, participants were also surveyed for previous history of lower extremity overuse injuries and stress fractures. Any medical illness and medications were documented.
Statistical Analysis: Data were analyzed with IBM SPSS statistics software version 16.0 (IBM Corporation, New York, USA). The descriptive analysis of the data was expressed as mean, SD, frequency and percentage. Statistical comparisons of means were calculated using a student’s t test. For multivariate analysis, a multiple logistic regression analysis was performed, with stress fractures as the dependent variable and the assessed risk factors as independent variables. P<0.05 was considered statistically significant.
Of the 108 female military recruits initially enrolled in this study, 8 were excluded from this study as they expelled or withdrew form the basic training program for reasons that were unrelated to this study. Participants who were excluded from the study were also excluded form statistical analyses. Basic and demographic characteristics of the 100 recruits that successfully completed the study are shown in Table 1.
During the 6-month of basic training program, 17 of the 100 participants (17%) were referred to the orthopedic clinic with the following complaints: 5 with knee pain (5%), 2 with hip pain (2%), 8 with leg pain (8%) and 2 with ankle pain (2%). A total of 8 participants were diagnosed with stress fractures (8%). Stress fractures of the tibia were most common (n=7, 87.5%), followed by femur (n=1, 12.5%). Of the 8 participants sustaining stress fractures, 5 had bilateral stress fractures (62.5%) and 3 had unilateral stress fractures (37.5%).
Mean and SD of the selected demographic and physical characteristics were calculated using a student’s t test and presented in Table 2 according to stress fracture status. A logistic regression analysis was performed to assess the effects of age, weight, BMI, height, hemoglobin, ALP, calcium, vitamin D, age of menarche, marital status, OCP use and menstrual period on stress fracture (Table 3). The model explained 70.4% of variance of stress fracture. Married women were 1.428 times more likely to exhibit stress fracture than single women. Women with irregular periods had a higher risk for developing a stress fracture (Figure 3).
Although non-modifiable risk factors for stress fractures cannot be altered, their importance lies in identifying individuals at higher risks of injury [8]. Females have been shown to have a significantly higher risk of sustaining stress fractures when compared with males [2]. During 13 weeks of US Marine Corps basic training, 5% to 7% of female recruits sustained stress fractures [9,10]. On the other hand, an incidence of 3% was reported in a sample of male recruits from the US Marine Corps 12-weeks basic training [11]. Similarly, during UK Army recruits basic training; stress fracture incidence was 10.9% in females versus 3% in males [12].
Menstrual irregularities and age at menarche have been previously associated with stress fractures. We found that age of menarche was strongly associated with risk of stress fracture. Females who had earlier ages of menarche were significantly more likely to develop a stress fracture (p<0.05). Contrarily, Tenforde, et al. reported a higher risk of stress fracture in females with late menarche (15 years or older, p=0.01) [13]. Moreover, Australian female athletes who suffered from stress fractures all had a later age of menarche and fewer menstrual cycles/year, compared to controls [14,15]. A review of 207 female collegiate athletes found a stress fracture incidence 3.3 times higher in those with irregular menses [16]. In the US Army, a survey of 1630 women showed that those with amenorrhea of more than 6 months experienced one or more stress fractures [17].
Ethnicity is another non-modifiable risk factor for stress fractures. Multiethnic studies of the American Army reported significantly higher risks for stress fractures in Caucasians, when compared to blacks [17-19]. Furthermore, in spite of their lower serum vitamin D levels, black females have shown higher bone mineral density on dual energy x-ray absorptiometry compared to white females [20]. This means that bone quality and mineral density could be attributed to factors other than vitamin D levels. Hence, the decreased risk seen in Blacks [21]. Black individuals are believed to have adaptive responses to low vitamin D levels which are protective against skeletal injuries [20].
Calcium and Vitamin D are essential for bone health [22]. Inadequate levels of calcium and vitamin D are associated with an increased risk of osteoporosis, osteomalacia, and stress fractures [22-24]. In a randomized double-blind placebo-control study of 5201 female Navy recruits during 8 weeks of basic training, Lappe, et al. found that subjects receiving calcium and vitamin D supplementation had a 20% lower incidence of stress fractures compared to controls receiving placebo [23]. However, in our study, 21% and 78% of participants had vitamin D sufficiency and deficiency, respectively. This endemic of hypovitaminosis D is in lieu with published literature on vitamin D levels in Arabs [25-27]. However, with such high prevalences of hypovitaminosis D in Arab populations, and because vitamin D was not shown to be a significant risk factor to stress fractures in our study population, we hypothesize an adaptive response present in Arabs, similar to that seen in Blacks which warrants further investigations [20,21]. Moreover, the high prevalence of hypovitaminosis D seen in Arabs could be attributed to cultural conservative clothing and decreased outdoors activities; sun exposure, due to high temperatures in Arab countries [28]. Nevertheless, low vitamin D levels were also reported in Arab-American females living in Michigan which again emphasizes the need for further investigations [29].
In analyzing stress fractures in our population, the tibia was the most common injury site (87.5%), followed by the neck of femur (12.5%). This is in accordance with published literature. Armstrong, et al. reported that 50% of stress fractures occurred in the tibia. Of all stress fractures, the tibia was the commonest site in males (72.2%) and females (100%) [30]. Similarly, in a study of US Marine Corps, stress fractures of the tibia represented 71% of stress fractures [31]. In investigating the biomechanical factors associated with stress fractures, Milner, et al. conducted a large cohort study of female runners over 4 years. They noticed that the stress fracture group had increased average vertical loading forces acting on their tibias. This was due to their knees having a greater valgus angle, internal rotation and sagittal knee stiffness, compared to others [32,33]. Moreover, foot structure, knee kinematics and biomechanical movement patterns have been associated with stress fractures and their locations [34].
Recently, genetic variations in vitamin D receptors (VDR) have been associated with stress fractures. Fokl and Bsml polymorphisms of the VDR gene can decrease bone content and have been shown to increase the risk of stress fractures in Caucasians and Spanish individuals, amongst other populations [35-37]. He, et al. found that vitamin D receptor gene variations influence bone mineral density and osteoporosis [38]. Genetic variations in vitamin D receptors in Arabs have not been previously investigated. The high prevalence of hypotivaminosis D found in Arabs warrants future studies into the presence of specific genetic variations in vitamin D receptors.
We found that weight and BMI were both associated with stress fractures. Females with higher weights and BMIs were more likely to develop stress fractures. This is contrary to Tenforde, et al. who reported that females with low BMI (<19) were at three times greater risk of sustaining a stress fracture (p<0.05) [13]. A higher BMI was also shown to be protective against the development of stress fractures in females during BCT [2]. Thinner women and females losing weight have been shown to exhibit more rapid bone loss and decreased bone mass density (BMD) which is the rationale behind such results [39]. Nevertheless, mechanical loading is an essential stimulus to bone formation through increasing osteoblasts and osteoclasts differentiation [40]. A high BMI can either be due to lean mass or adiposity, in which each has its influence on bone. It is well established that dynamic load exerted from muscle contraction is more anabolic to bone than static load [41]. Also, skeletal unloading from immobilization and inactivity was found to increase adipocyte differentiation and inhibit osteoblastic differentiation [42]. Furthermore, higher adiposity is highly associated with decreasing the rate of bone formation [43]. Finally, research has shown that in attempting to reduce weight, caloric restriction exerts a harmful effect to bone health whilst exercise plays a protective role through stimulating bone formation [44,45]. In summary, obesity on its own does not provide mechanical advantage to bone if not accompanied by greater lean mass and physical activity. Therefore, body composition would probably be a more specific tool than BMI in evaluating stress fractures risk. Although we found that females with higher BMI were at higher risks of developing stress fractures, contradicting previous studies, this rationale serves as a valid explanation, which warrants further investigations.
The most important limitation to our study is the relatively small sample size. Due to such small sample size, the risk factors for stress fractures in female Arabs could not be fully appreciated. Nevertheless, our preliminary data suggests that risk factors for stress fractures in female Arabs might differ from those of other ethnic populations. Therefore, we recommend the study of these risk factors in larger populations.
Identifying risk factors for stress fractures is important in attempting to reduce their incidence. Risk factors for stress fractures vary between different ethnic populations. This is the first preliminary study to investigate risk factors for stress fractures in Arabs, and could serve as a basis for future stress fracture research in the region.
We would like to thank Maj. Gen. Prof. Khalid Bin Ali Al-Khalifa for his continuous support and encouragement towards research at the Royal Medical Services, Bahrain Defence Force hospital. We thank the medical battalion at the Royal Medical Services for their interests and inputs in our research project. Last, but not least, we thank Maj. (Dr.) Ahmed AlAnsari and Ms. Aaruni Suresh for statistical support.
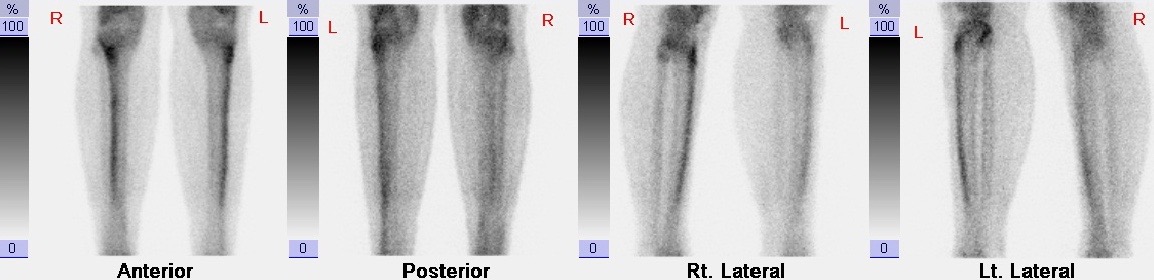 |
Figure 1: SPECT bone scan image showing increased uptake over the tibial shafts consistent with shin splints |
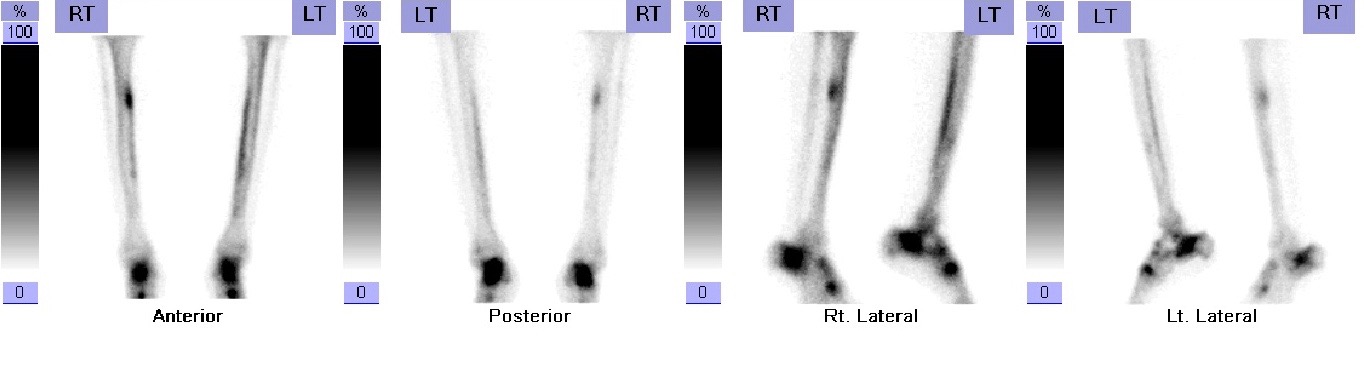 |
Figure 2: SPECT bone scan images showing stress fracture of the right tibial shaft |
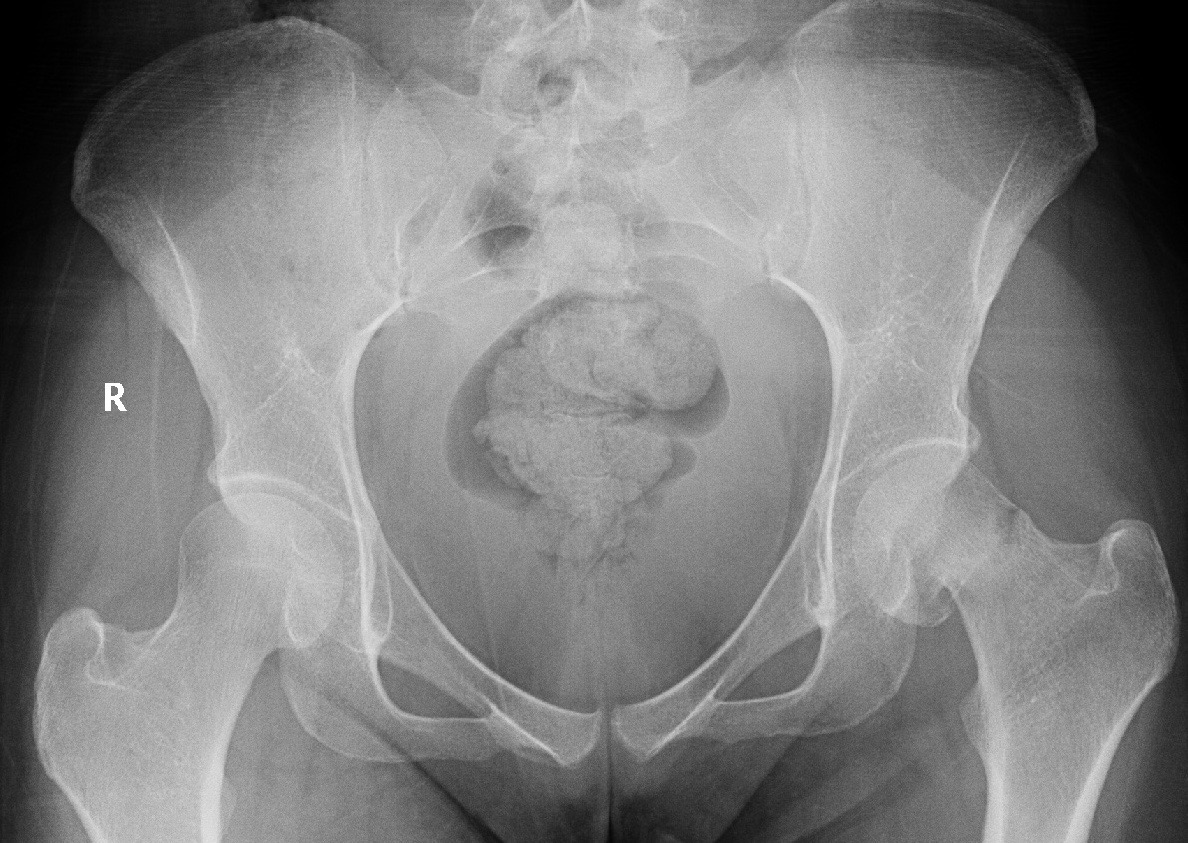 |
Figure 3: Plane radiograph of the pelvis and both hips showing stress fracture of the left neck of femur with a varus deformity |
Variables |
Mean ±SD |
|---|---|
Age |
22.13 ± 2.80 |
Weight |
59.79 ± 8.82 |
Height |
158.45 ± 5.0 |
Body Mass Index (kg/m2) |
23.81 ± 3.31 |
Haemoglobin (g/dL) |
11.88 ± 1.06 |
Alkaline Phosphatase (IU/L) |
77.53 ± 18.08 |
Vitamin D (ng/ml) |
26.87 ± 23.80 |
Menarche (years) |
13.13 ± 1.38 |
Time since Menarche (years) |
9.0 ± 0.299 |
Marital status n (%) |
|
Single |
75 (75.0) |
Married |
25 (25.0) |
Period, n (%) |
|
Regular |
88 (88.0) |
Irregular |
12 (12.0) |
Oral Contraceptives n (%) |
|
Yes |
3 (3.0) |
No |
97 (97.0) |
| Table 1: The demographic characteristics of the sample population (N=100) | |
No Stress Fracture |
Stress fracture |
|||||
|---|---|---|---|---|---|---|
N |
Mean |
Std. Deviation |
N |
Mean |
Std. Deviation |
|
Age |
92 |
22.0 |
2.70325 |
8 |
23.5 |
3.77964 |
Weight (Kg)* |
92 |
58.8 |
8.27273 |
8 |
71.0 |
7.40656 |
Body Mass Index (kg/m2) |
92 |
23.4 |
3.02547 |
8 |
29.0 |
1.84303 |
Height (cm) |
92 |
158.6 |
5.05946 |
8 |
156.4 |
3.88909 |
Haemoglobin (g/dL) |
92 |
11.9 |
1.08225 |
8 |
11.9 |
0.86839 |
Alkaline Phosphatase (IU/L) |
92 |
77.7 |
17.78191 |
8 |
75.8 |
22.62584 |
Vitamin D (ng/ml) |
92 |
27.7 |
24.75216 |
8 |
17.5 |
4.93644 |
Menarche* |
92 |
13.2 |
1.39722 |
8 |
12.1 |
0.64087 |
* p<0.05 (Statistically significant) |
||||||
Variable |
B |
S.E. |
Wald |
df |
Sig. |
Exp(B) |
95% C.I.for EXP(B) |
|
|---|---|---|---|---|---|---|---|---|
Lower |
Upper |
|||||||
Age |
0.16 |
0.34 |
0.23 |
1 |
0.63 |
1.17 |
0.61 |
2.27 |
Weight |
0.29 |
2.37 |
0.015 |
1 |
0.90 |
1.34 |
0.01 |
139.77 |
BMI |
0.00 |
5.73 |
0.00 |
1 |
1.00 |
1.00 |
0.00 |
75651.55 |
Height |
-0.57 |
2.07 |
0.07 |
1 |
0.78 |
0.57 |
0.01 |
33.05 |
Haemoglobin |
0.68 |
0.64 |
1.13 |
1 |
0.29 |
1.97 |
0.56 |
6.90 |
Alkaline Phosphatase |
0.04 |
0.06 |
0.43 |
1 |
0.51 |
1.04 |
0.92 |
1.17 |
Calcium |
-1.54 |
6.50 |
0.06 |
1 |
0.81 |
0.22 |
0.00 |
73582.15 |
Vitamin D |
-0.17 |
0.24 |
6.52 |
1 |
0.47 |
0.84 |
0.52 |
1.35 |
Menarche |
-1.17 |
0.98 |
1.41 |
1 |
0.23 |
0.31 |
0.05 |
2.14 |
Marital status |
0.36 |
1.45 |
0.06 |
1 |
0.81 |
1.43 |
0.08 |
24.70 |
OCP use |
-0.80 |
4.52 |
0.03 |
1 |
0.86 |
0.45 |
0.00 |
3183.94 |
Period |
2.54 |
1.59 |
2.56 |
1 |
0.11 |
12.69 |
0.57 |
284.17 |
Constant |
73.12 |
327.67 |
0.05 |
1 |
0.82 |
56.56 |
||
Table 3: Stress fracture incidence and OR by demographic, physical body structure |
||||||||






































