Top Links
Journal of Obesity and Overweight
ISSN: 2455-7633
Adipogenesis in Obesity is Modulated by IP6 in Peanuts through Activation of the Nuclear Receptors (PPARs)
Copyright: © 2016 Malarvizhi R. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Related article at Pubmed, Google Scholar
Increased life style changes have led to global epidemic of obesity. The global prevalence of obesity protracts to increase, with devastating consequences for overall health. Thus by exploring novel beneficial properties of natural products and their scientific indications are the need of the hour and can bring about a whole new revolution in nutraceutical industries and health care sectors. IP6 (Inositol hexaphosphate) is one of the potent bioactive molecule in peanuts and in many cereals, grains, nuts, legumes and oil seeds. IP6 is a metal chelator and may interact with minerals, proteins and starch and thus alters the solubility, functionality, digestion and absorption of the food components. The present study warrants to quantify IP6, a potent metal chelator present in peanuts a commonly consumed food supplement in India and to identify its role as an anti-obese molecule in 3T3-L1 cells. The content of IP6 in raw, boiled and roasted peanuts was quantified by high performance liquid chromatography (HPLC) to identify the presence of IP6 in the various processing methods. IP6 content in raw peanuts was high when compared to roasted and boiled peanuts. In order to study the influence of IP6 on 3T3-L1 cells the Oil Red O staining of the adipocytes were done, which showed a dose dependent inflation in the lipid accumulation thereby reducing the circulating lipids which are modulated by the action of the Peroxisome Proliferator-Activated Nuclear Receptors (PPARs). Docking studies revealed that the binding energy of IP6 with peroxisome proliferator-activated nuclear receptor gamma (PPARγ) is higher than peroxisome proliferator-activated nuclear receptor alpha (PPARα). This was further confirmed by gene expression studies using real time-PCR. However, further studies are warranted in other target genes which mediate pathologies associated to obesity.
Keywords: Obesity; IP6; Peanuts; PPARs; 3T3-L1; Adipogenesis
Abbreviations: IP6: Inositol hexaphosphate; DMEM: Dulbecco’s Modified Eagles Medium; MTT: 3-(4, 5-dimethylthiazol-2-yl)-2, 5-diphenyltetrazolium bromide, PDB: Protein Data Bank; RID: Refractive Index Detector; TG: Triglycerides; PPARs: Peroxisome Proliferator-Activated Nuclear Receptors; PEPCK: Phosphoenolpyruvate Carboxykinase; PCR: Polymerase Chain Reaction
Obesity continues to increase around the world, with its devastating effect on the overall health of man. It is defined as accumulation of adipose tissue in excess so as to impair the health of an individual [1]. Obesity is not a single disease but a miscellaneous group of conditions with numerous cause each of which is eventually expressed as obese phenotype [2]. It may also be caused due to other contributing factors such as genetic, environmental, physiological and psycho-social impacts [3]. Globally, nearly 3.4 million individuals die due to obesity [4]. Further, it is considered to be the 5th major risk factor accounting for global death [5]. Hence, it is rightly termed as a killer disease [6]. Adipogenesis the root cause for obesity is the process of conversion of pre-adipocyte into mature adipocyte involving a two-step process. During this process there is a swap in its morphology and associated gene expression [7]. Furthermore, adipose tissue pivotal in lipid and glucose metabolism besides producing substantial number of hormones and cytokines exhibits both exocrine and endocrine functions [8]. It has an impact over the various vital functions such as appetite, immunological responses, inflammatory responses, angiogenesis, blood pressure regulation and reproductive function [9].
Food and Nutrition Science has highlighted the possibility of modulating specific physiological function in the organism through food intake [10]. Thus with the effective use of food we can combat various diseases as emphasised by the father of medicine “Let food be thy medicine, thy medicine shall be thy food”. Moreover, nowadays attention of people has been shifted to drugs of natural origin which exhibit least side effects. The role of food in modulating the physiological functions is mediated by the phytochemicals. It is interesting to note that adipogenesis is inhibited by a profuse number of phytochemicals present in food [11]. Hence, off late research groups around the globe have directed their research in identifying the mechanism of phytochemicals on adipogenesis [12,13].
Arachis hypogaea commonly called as peanut or groundnut is consumed as a food product as it is a rich source of energy. Apart from the high amounts of mono and polyunsaturated fatty acids in it, presence of bioactive molecules including plant sterols, essential amino acids, micronutrients and phytochemicals such as resveratrol, IP6, coumaric acid, dehydrated quercetin are appreciable [14]. As whole or individual phytoconstituents they directly or indirectly mitigate dyslipidaemia [15], obesity [16,17] and modify postprandial oxidative stress [18]. IP6 otherwise known as phytate or phytic acid [19] exhibits potential therapeutic action on diabetes mellitus [20], urolithiasis [21], dental caries [22] and a whole range of cancers [23]. However, there are scanty studies on the role of IP6 in peanuts on adipogenesis till date. In the present study the quantity of IP6 in raw, boiled and roasted peanuts was measured using HPLC and the possible mechanism of action of IP6 on adipocyte differentiation was studied by in-vitro and in-silico analysis.
Purified IP6 is a gift sample from Resveratrol Partners, USA for the study. Raw peanuts [Pondicherry variety VR-1] were purchased from the local agricultural station for the analysis of IP6 in peanut. All cell growth and culture media used were of tissue culture grade and purchased from HiMedia. MTT [3-(4,5-dimethylthiazol-2-yl)-2,5 diphenyltetrazolium bromide] and Isobutylmethylxanthine (IBMX) was purchased from Sigma-Aldrich, USA. All other chemicals and reagents used were of analytical grade, SRL Ranbaxy, India. Glucose estimation kit was purchased from Accurex biomedical Pvt Ltd, India.
Extraction of phytic acid from peanut was done in three different processed peanuts such as raw, boiled and roasted peanuts [24]. The powdered peanuts in the above conditions were defatted overnight with acetone. Acetone was removed by centrifugation at 15,000 rpm for 5 minutes. Supernatant obtained was discarded and the pellet was dried over filter paper. The dried sample was extracted with 2.4% hydrochloric acid (HCl) for 1-3hr [25]. Sample extracts of 2.4% HCl are boiled with 0.03% Ferric chloride hexahydrate to aid in preparation of ferric phytate. Subsequently; ferric phytate was washed and converted to soluble sodium phytate and insoluble ferric hydroxide precipitate with the addition of 1.5N sodium hydroxide and boiled it for 30 minutes. The soluble sodium phytate was made to known volumes and an aliquot was then injected into C18 reverse phase HPLC column for the quantification of IP6. A 5mM sodium acetate solution was used as a mobile phase at a flow rate of 0.5ml/minute [26].
The 3T3-L1 pre-adipocytes were cultured in Dulbecco’s Modified Eagles Medium (DMEM) enriched with 10% (v/v) fetal calf serum, 4.5g/dl glucose; 4mM stable glutamine, 50k IU/l penicillin and 0.05g/l antibiotic streptomycin at 37 oC in a 5% CO2 cell incubator. Cells were sub-cultured for every two days using 0.25% (w/v) Trypsin- EDTA solution and split into 1:3 ratio into fresh DMEM medium. Cells were sub-cultured when they reach up to 80% confluent stage.
3T3-L1 mouse pre-adipocytes cell line was procured from National Centre for Cell Sciences (NCCS) Pune, India. The viability of 3T3-L1 cells after treatment with various concentrations of pure IP6 was assayed by the reduction of 3-(4, 5-dimethylthiazol-2-yl)-2, 5-diphenyltetrazolium bromide (MTT) to formazon insoluble dye [27]. Briefly cells were seeded into a 96- well micro titer plate (500-10,000 cells in 200μl media) per well and allowed to reach at confluent stage. The cells were incubated at 37 oC with 5% CO2 overnight to allow the cells to attach to the wells and multiply. Subsequently 2μl of different concentrations of IP6 to each well was added and incubated. After 24hrs, cells were washed with PBS and 50μl of MTT (5mg/ml) was added followed by incubation for 4 hrs at 37 oC under dark conditions. Thereafter, media was aspirated and the formazan dye crystals formed were re-suspended in 200 μl of DMSO and thoroughly mixed. The absorbance read at 570 nm using a microplate reader (BIORAD) was directly correlated to the quantity of cells.
The cells were seeded at a concentration of 0.2x105 cells per well in a 24-well plate and allowed to reach 80% confluence. After two days of post confluent stage, cells were stimulated with differentiation medium or MDI cocktail (DMEM, 10% FBS, 1μM Dexamethasone, 0.5mM Isobutyl methyl xanthine (IBMX) and 10μg/ml insulin) for 3 days. After 3 days, the induction media was replaced with adipocyte maintenance media (10% FBS/DMEM with 10 μg/ml insulin). Thereafter, media was changed for every two days with the adipocyte maintenance media. Role of IP6 on the process of conversion of pre-adipocyte to adipocyte differentiation was studied by incubating the pre-adipocyte cells with 2μl of test drug (pure IP6) in various concentrations. The extent of differentiation was quantified by staining the cells with Oil Red O (ORO). To stain the lipids stored in adipocytes, the media was aspirated from the wells, washed with 10% formalin PBS and then incubated with fresh formalin PBS for 1 hour to fix the cells. Formalin was removed, washed with 60% isopropanol and air dried completely. Fixed cells were then stained with Oil Red O solution for 10 min and washed with water 4 times to discard the unbound stain. The appearances of stained cells were then captured on a digitalized microscope. Cells were completely air dried to elute ORO by using 100% isopropanol. The isopropanol with ORO was aspirated up and down several times for efficient solubilisation of ORO. The intensity of ORO in isopropanol was measured at 500 nm with 100% isopropanol as blank.
The cells were differentiated into adipocytes in 24-well plates. Well differentiated adipocytes were serum starved with DMEM containing 0.2% BSA for 24 hours and then co-incubated with different concentrations of pure IP6 for 24 hours. The medium was then extracted to estimate the glucose levels by the glucose oxidase method. The amount of glucose consumption was calculated by the difference in the glucose concentration of the blank wells and the cell-plated wells [28]. Results were expressed in mM glucose consumption by comparing with untreated controls.
Molecular docking studies were done for IP6 with PPAR α and PPAR γ. The protein receptor, PPARα (PDB ID- 4BCR) and PPARγ (PDB ID-4HEE) were retrieved from Protein data bank (PDB) (www.pdb.org/pdb). Using Pymol the structures of the receptors were prepared for docking by removing the co-crystallized ligand and additional water molecules to make it as a nascent receptor. Ligand structures were generated by Chemsketch (www.acdlabs.com) in mol format and the generated structures were converted into Pdb format using Chimera (https://www.cgl.ucsf.edu/chimera). Subsequently the generated Pdb structures were used for docking by using the flexible docking protocol, Auto dock (Auto dock tools- 1.5.4 version).
3T3-L1 pre-adipocytes were grown in T-75 flasks and allowed to get confluent. Cells were pre-incubated with IP6 (5ng/ml) for 6 hrs followed by differentiation with MDI media. Total RNA was isolated from 3T3-L1 cell lines by using Trizol reagent. RNA was quantified using a Nano drop and cDNA (complementary DNA) was synthesised using a commercial kit (high cDNA, Applied Biosystem). Quantitative real-time PCR (q-RT PCR) was performed in AbiPrism™ 7500 sequence detection system (Applied Biosystems). Gene-specific primers are as follows PPARα- F 5’-GCC ATC TTC ACG ATG CTG TCC TCC-3’ R 5’-GTA GAT CTC TTG CAA CAG TGG GTG C-3’, PPARγ- F 5’-GGA TTC ATG ACC AGG GAG TTC CTC-3’ R 5’-GCG GTC TCC ACT GAG AAT AAT GAC-3’, and β actin- F 5’-GGC CAA CCG TGA AAA GAT G-3’ R 5’-GGA TCT TCA TGA GGT AGT CTG TC-3’.Specific genes were amplified in a reaction mixture containing 1μl of template (cDNA), 300nM of forward and reverse primers and 1x SYBR green PCR master mix and the reaction was performed as per the kit manufacturer’s instruction (Applied Biosystems). The change in fluorescence of SYBR green dye in every cycle was monitored, and threshold cycle (ct) above background for each reaction was calculated.
The results were expressed as mean ± SEM and analysed using one way ANOVA by SPSS software version 20.
The present study involves the work of analysing the content of IP6 a metal chelator, largely present in peanuts and was estimated in the raw, boiled and roasted peanuts. The IP6 recovered by acid precipitation was estimated by HPLC using Refractive Index Detector (RID). Various methods are available for the determination of IP6 and it was reviewed by Harland BF [25]. However a reverse phase HPLC analysis of IP6 was done following the methodology of Reddy NR and co-workers [26]. IP6 elutes immediately after the void volume, which corresponds to a retention time of 2.847. It is well separated with 5mM sodium acetate along with the IP6 minor refractive index peaks which are seen in all the three processed peanut sample chromatograms. A typical high-performance liquid chromatogram of phytic acid is shown in Figure 1. In the present study the phytic acid content in the raw sample is observed more than the roasted and boiled samples through the response of the RID in the HPLC. The amount of phytic acid present in raw, roasted and boiled peanut sample and their retention times are given in Table 1. The retention time of pure IP6 and IP6 derived from the processed peanut were same which confirmed the purity of the IP6 and was used for further analysis.
Cell viability assay for IP6 was done with various concentrations (10ng, 100ng, 1000ng, 5000ng, 10000ng, 20000ng, 40000ng, and 80000ng) by employing MTT assay on 3T3-L1 cell line. MTT assay is dependent on the conversion of MTT into formazan crystals by living cells [27]. The non-toxic dose of IP6 on 3T3-L1mouse pre-adipocytes was evidenced by the viable cells as shown in Figure 2. IP6 was found to be non-toxic till a concentration of 10000ng after that as the dose increases the cell viability decreases. Hence, the concentration within 10000ng of IP6 was used for further assays.
Oil red O is a lysochrome diazo dye used for staining lipids. To examine the extent of lipid accumulation caused by inhibition through differentiation, various concentrations of IP6 were incubated in the 3T3-L1 cells and accumulation of fat droplets was measured using lipid staining oil red O. Based on the results, IP6 induces lipid accumulation in mature 3T3-L1 adipocytes in a dose dependent manner which is represented in Figure 3. IP6 showed an upward tendency in accumulation of lipid which is directly proportional to its dose, thereby exhibiting maximum activity at a concentration of 10000ng. Microscopic image of triglycerides (TG) accumulation in 3T3-L1 cells is shown in Figure 4.
In the present study three different concentration of pure IP6 exhibits gradual increase in the glucose consumption with an increase in time and dose. The maximum glucose consumption activity is shown at the 24 hour of incubation adipocytes and it was shown in the Figure 5. The glucose consumption capacity was stimulated by IP6 in dose dependent manner.
The influence of IP6 on the peroxisome proliferator-activated nuclear receptors (PPARs) was done using in-silico analysis. The ligand binding pocket (LBP) of both PPARα and PPARγ was docked with an agonist IP6 by flexible docking protocol, Auto dock (Auto dock tools- 1.5.4 version) to identify the binding affinity. All the possible rotatable bonds of the agonist were considered in the docking process for analysing the best binding conformation with PPARα and PPARγ. In the crystal structure of PPARα (PDB ID- 4BCR), the LBP contains the amino acids Proline 238 and Phenylalanine 338. These amino acids were targeted during the grid preparation for docking. The ligand (IP6) was prepared based on the number of rotatable bonds and ten possible conformations were achieved wherein the seventh pose showed strong hydrogen bonding with Histidine 457 with a conformational binding energy of -5.67 as shown in Figure 6A. Serine 342 which is one of the active site amino acid of PPARγ (PDB ID-4HEE) actively docked to IP6 with a conformational binding energy of -10.39. The formation of hydrogen bonds between IP6 and the amino acids in the LBP of PPARγ is shown in Figure 6B. Thus in-silico docking studies reveal that PPARα and PPARγ shows positive interaction with IP6. However, it is interesting to know that the binding efficiency of IP6 with the isoform PPARγ is higher than that of PPARα probably emphasizing the mode of action too.
To explore the mechanism underlying suppression of adipocyte differentiation by IP6 in modulating obesity associated conditions, the mRNA levels of genes such as PPAR α and PPARγ, involved in lipogenesis and lipolysis mediated by adipocytes were examined using quantitative real time-PCR. Figure 7 represents the expression of PPARα and PPARγ level on the treatment with and without IP6 compared with adipocyte. PPARα and PPARγ shows the upregulation of gene expression with the treatment of IP6 at a maximum of 1.5 and 6.4 fold increases is observed respectively.
Obesity prevalence is increasing worldwide with various associated diseases such as cardiovascular diseases, diabetes and hyperlipidaemia [29]. Most of the management strategies used for treating obesity cause several adverse effects which prompts for an alternative therapy. Therefore in the present study the impact of IP6 a bioactive compound present in peanuts consumed commonly was studied on adipocytes (3T3-L1 cell line) to study its role in adipogenesis. Scientific evaluation of the unique properties of the natural food products that are immensely beneficial to the human population can bring about a revolution in the food industries and the health care sectors. Peanut, an easily available legume, is being consumed by vast majority of the people around the world. It possess intrinsic medicinal properties such as anti- inflammatory, antioxidant, anticancer and antifungal property and serves as a natural boon in saving the health of the people [30]. Hence, peanuts have recently been attracted as a functional food [31]. As per the HPLC analysis, the IP6 content in raw peanuts is higher when compared to roasted and boiled peanuts. Previous study on the content of phytate in whole wheat [32], rice bran, sesame seed [33] and beans reveals that IP6 is considerably high in rice bran and sesame seeds. Levels of phytic acid can vary with growing conditions [34]. However, based on the results obtained in the present study on the three types of processed peanuts raw peanuts which contain highest content of IP6 (7.09 mg/g) is considered less suitable for consumption whereas boiled peanuts contain only (0.005mg/g) of IP6 and exhibit minimal bioactivity. IP6 a metal chelator is required in minute quantities, since it might chelate the necessary mineral elements required for general physiological function of the body. Hence, it is recommended that roasted peanuts may be beneficial with moderate level of IP6 (2.49mg/g) compared to raw and boiled peanuts.
Glucose the main source of metabolic fuel in mammalian cells and its receptors inherently are present in the cells and facilitate the free passage of glucose in the cells. Glut-4 helps in blood glucose clearance and it is highly expressed in tissues like skeletal muscle, heart muscle and fat [35]. In the present study three different concentration of IP6 exhibits gradual increase in the glucose content with an increase in time probably mediated by the Glut-4 transporter. Further, incubation of adipocyte with IP6 shows the maximum glucose consumption in a dose and time dependent manner. The regulation of glucose homeostasis depends on hepatic glucokinase enzyme [36]. The phytic acid coupled with enhancement of glucokinase activity plays a vital role in anti-hyperglycaemic action and inhibition of Glucose-6- phosphatase and Phosphoenolpyruvate carboxykinase (PEPCK) in the liver. Higher levels of hepatic glucokinase tend to increase the blood glucose utilisation level for energy production or glycogen storage in the liver thereby resulting in reduced blood glucose level [37]. The results obtained by in-vitro assay in this study are in line with earlier studies and is worth investigating for its mechanism of action in in-vivo studies too.
Adipocyte the basic factor in causing obesity is found to be present in large quantity in white adipose tissue [38]. The classical perception of adipocytes as storage depot of free fatty acids has been replaced by the notion that adipocytes have a major role to play in lipid and glucose metabolism and in the secretion of several hormones and cytokines eg; angiotensinogen, TNF-α, IL-6, adiponectin and leptin [39,40]. Based on adipocyte differentiation assay IP6 showed an upward tendency in accumulation of lipid which is directly proportional to its dose, thereby exhibiting maximum activity at a concentration of 10000ng. Hypocholesterolemic effect possessed by IP6 could act as potential source in the clinical management of hyperlipidaemia and diabetes [41]. In an earlier study Sangeetha et al., showed the adipogenic property of diosgenin by reducing the circulating lipids and related it to be mediated by PPARγ [42]. A similar result is seen in the present study and hence substantiates that IP6 enhances lipid accumulation in the adipocytes thereby reducing the circulating lipids. Moreover, agonists have a capacity to mitigate the circulating free fatty acids by lipolysis process [43]. This might probably mitigate cardiovascular disorders associated with obesity.
Peroxisome proliferator-activated receptors (PPARs) belonging to the nuclear receptor superfamily with 3 isotypes namely PPARα, β and γ, play a major role in the regulation of lipid and glucose metabolism [44]. Its activity is associated with the expression of several proteins involved in cell division, differentiation, growth, and also in energy pathway [45]. PPARα regulates liver and skeletal muscle lipid metabolism and glucose homeostasis [46]. As per in-silico studies the ligand (IP6) was docked with PPARα and it shows a conformational binding energy of -5.67. Crystallographic studies propose that ligand binding to PPARα induces a global balance of the receptor conformation which leads to an equilibrium of the PPARα ligand to a predefined structure, and enables protein-protein interactions with co-activators (agonist bound PPARα) or co repressors (antagonist-bound PPARα) [47]. PPARγ is primarily expressed in adipocytes where it plays a major role in the regulation of adipocyte differentiation, lipid uptake and adipogenesis of fat cells. PPARγ is a factor binding to a fat-specific enhancer of the ap2 gene which encodes an adipose-specific fatty acid binding protein [48]. In-silico docking studies revealed that IP6 was found to be actively bound with PPARγ and showed a conformational binding energy of -10.39. It is interesting to note that the binding efficiency of IP6 with, PPARγ is higher than PPARα probably due to selective interaction. In order to substantiate our in-silico results the expression of PPARα and PPAR γ was checked on 3T3-L1 cells. As seen in Figure 7, the expression of PPARα and PPAR γ was modulated as evidenced by the fold change of 6.4 fold in the case of PPAR γ and 1.5 fold of PPARα. It is interesting to note that IP6 interacts with both isoforms but at varied intensities and can be considered as a selective agonist for PPARγ compared to PPAR α thereby making it a potential lead for obesity associated metabolic syndrome. This is substantiated by the specific role of lipid accumulation in the adipocytes thereby reducing circulating lipids. Conversely, the glucose uptake is increased in the adipocytes mediated by PPARγ thereby increasing insulin sensitivity.
World health organization (WHO) has announced obesity as a disease in need for treatment. Results of the present work show the inherent capacity of IP6 in peanuts against obesity. The varied IP6 content in the raw, roasted and boiled peanuts infers that the content of IP6 in roasted peanuts is recommended for consumption. Further IP6 influences the glucose consumption and lipid accumulation in the adipocytes mediated by the isoforms of PPARs which was also confirmed both by gene expression and docking studies. However, further studies are warranted in experimental animals and with other target genes which mediate obesity associated pathologies to consider IP6 as a potential lead for obesity associated metabolic syndrome.
The authors wish to thank the infrastructure facilities developed by DST- FIST and UGC-SAP in the Department of Biotechnology, Pondicherry University for executing the present study. We also thank Bill Sardi of Resveratrol Partners, USA for the gift sample of IP6.
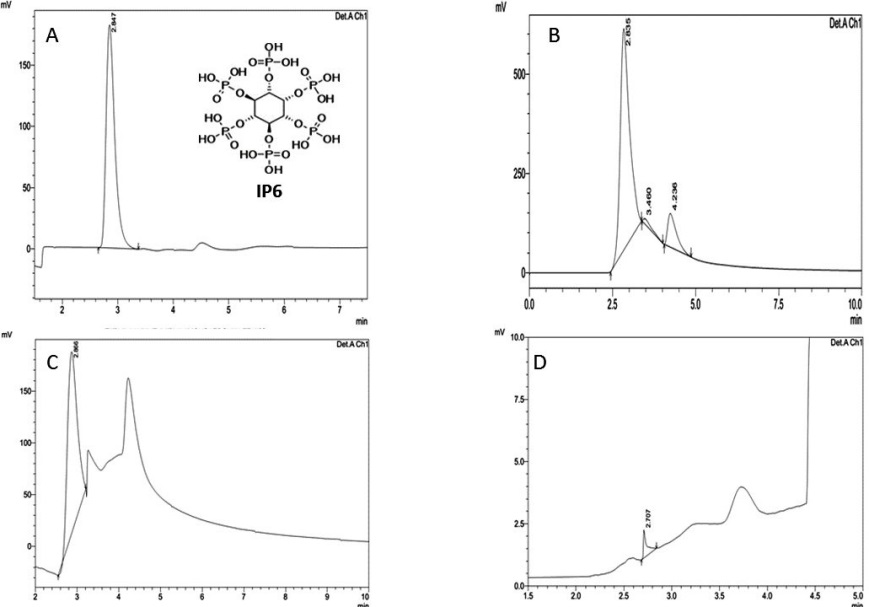 |
| Note: A) HPLC Chromatogram of Pure Inositol hexaphosphate (IP6); B) HPLC Chromatogram of Raw Peanut sample; C) HPLC Chromatogram of Roasted Peanut Sample; D) HPLC Chromatogram of Boiled Peanut Sample Figure 1: HPLC chromatogram of IP6 in Peanuts |
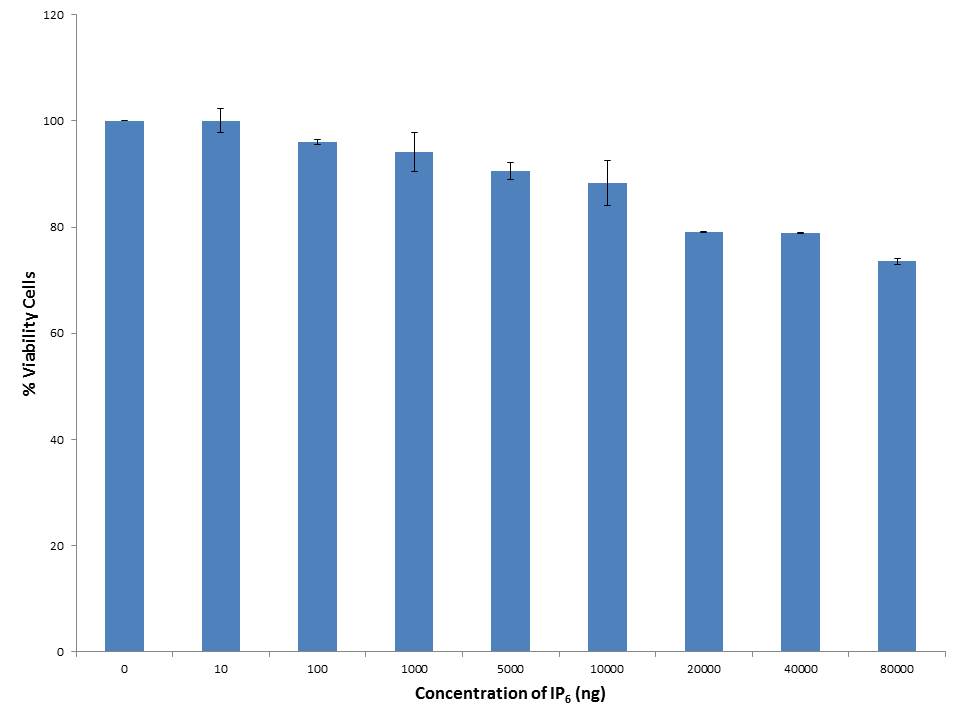 |
| Note: Results are expressed as Mean+SEM n = 3 Figure 2: Cell Viability assay on 3T3-L1 pre-adipocytes |
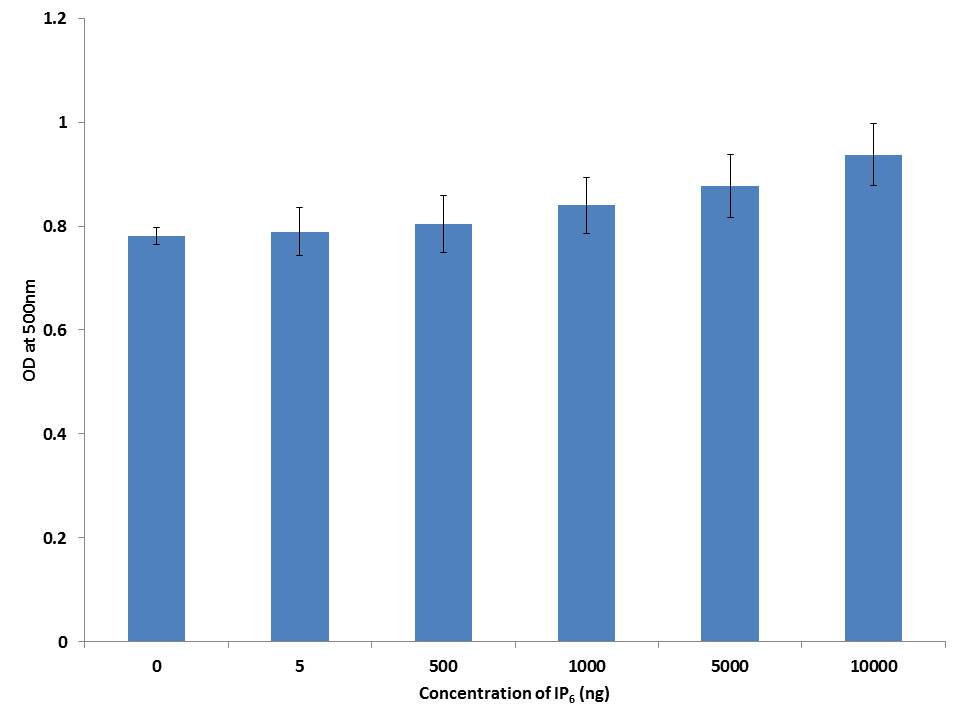 |
| Note: 3T3-L1 cells were stained with Oil red O stain to identify TG accumulation. Data reflects the Mean+SEM n = 3 Figure 3: Adipocyte differentiation assay |
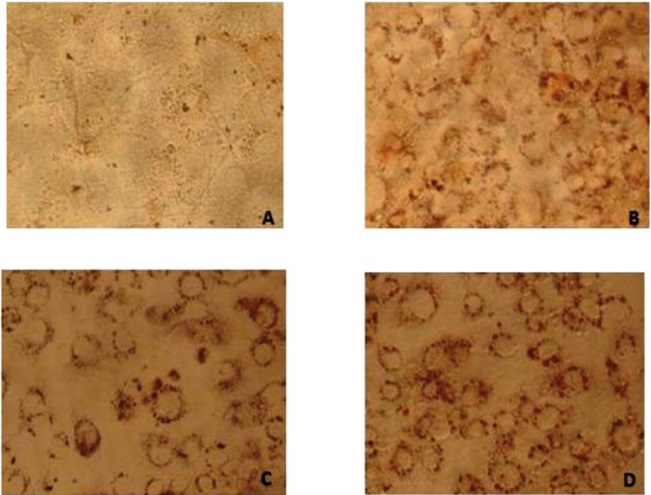 |
| Note: Digitalized under 20X; A) Pre-adipocyte staining with Oil Red O, showing no lipid accumulation in the cells; B) Adipocytes without drug treatment showing lipid droplets around the nucleus due to adipocyte differentiation; C) Stained cells showing lipid droplets in red colour was treated with IP6 (5ng); D) Stained cells showing lipid droplets in red colour was treated with IP6 (10000ng) Figure 4: HPLC chromatogram of IP6 in Peanuts |
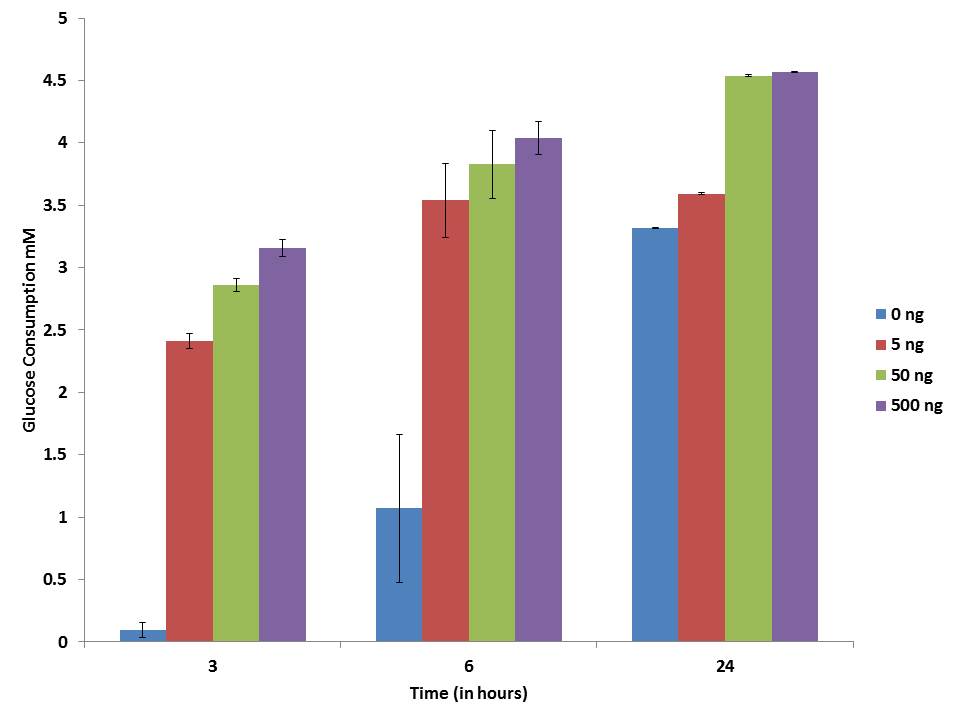 |
| Note: 3T3-L1 adipocyte cells incubated with different concentration of IP6 and its glucose level was measured at different time intervals Figure 5: Glucose consumption assay of Adipocytes |
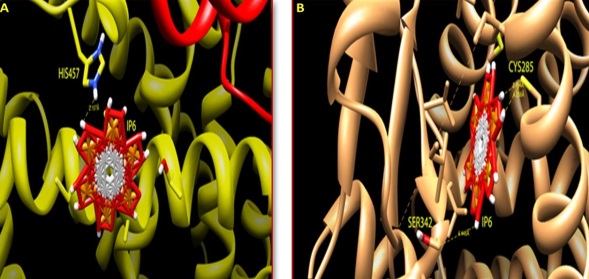 |
| Note:A) Agonist and interacting receptors was shown in stick mode, IP6 bound to the PPARα receptor protein by interacting with Histidine 457 amino acid residue; B) IP6 bound to the PPARγ receptor protein by interacting with Serine 342 amino acid residue Figure 6: Docking of PPARs with agonist IP6 |
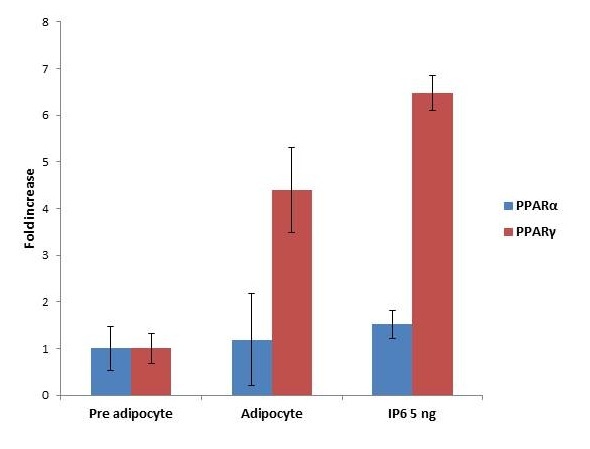 |
| Note: Impact of IP6 on PPARs expression. IP6 treated with 3T3-L1 cell lines shows the expression of PPARα and PPARγ in differentiated adipocytes. Results were plotted with Mean+SEM, n = 3 Figure 7: Expression of PPARs |
Sample |
Retention Time |
Area |
Height |
Quantity (mg/g) |
Raw Peanut |
2.866 |
2817094.0 |
175481.3 |
7.09 |
Roasted Peanut |
2.866 |
989995.1 |
70437.6 |
2.49 |
Boiled Peanuts |
2.707 |
2233.1 |
1116.8 |
0.005 |
Table 1: Quantity of IP6 in processed peanuts measured by HPLC |
||||






































